For oligodendrocytes—the central nervous system cells critical for brain function—age may not bring wisdom, but it does come with the power to cling to life for much, much longer than scientists knew. That’s according to a new study featured on the March 27 cover of the Journal of Neuroscience.
Mature oligodendrocytes took a shocking 45 days to die following a fatal trauma that killed younger cells within the expected 24 hours, Dartmouth researchers report. The findings suggest there’s a new pathway for efforts to reverse or prevent the damage that aging and diseases such as multiple sclerosis cause to these important cells.
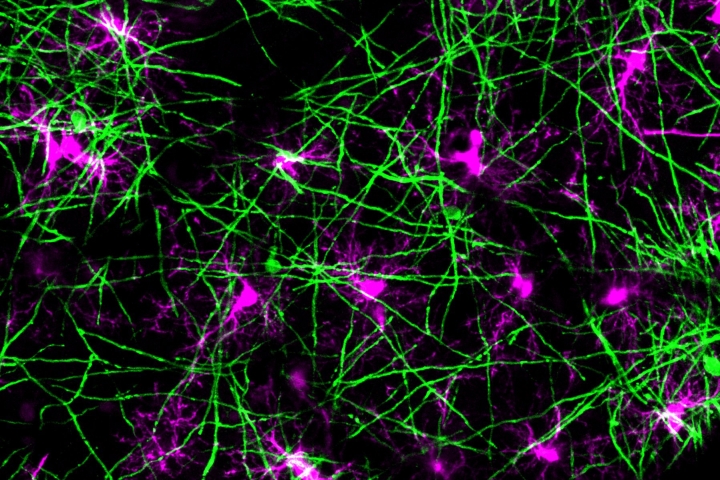
In the brain, oligodendrocytes wrap around the long, skinny connections between nerve cells known as axons, where they produce a lipid membrane called a myelin sheath that coats the axon. Axons transmit the electrical signals that nerve cells use to communicate; myelin sheaths—like the plastic coating on a copper wire—help these signals travel more efficiently.
Old age and neurodegenerative diseases like MS damage oligodendrocytes. When the cells die, their myelin production perishes with them, causing myelin sheaths to break down with nothing to replenish them. This can lead to the loss of motor function, feeling, and memory as neurons lose the ability to communicate.
Scientists have assumed that damaged oligodendrocytes—like all injured cells—initiate a cellular self-destruct called apoptosis in which the cells kill themselves. But Dartmouth researchers discovered that mature oligodendrocytes can experience an extended life before their death that has never been seen before. The findings pose the critical question of what in these cells changes as they mature that allows them to persist.
“We found that mature cells undertake a pathway that is still controlled, but not the classical programmed cell-death pathway,” says Robert Hill, an assistant professor of biological sciences and corresponding author of the paper.
“We think this is showing us what happens in brains as we age and revealing a lot about how these cells die in older people,” Hill says. “That unique mechanism is important for us to investigate further. We need to understand why these cells are following this pathway so we can potentially encourage or prevent it, depending on the disease context.”
First author Timothy Chapman, Guarini ’23, who led the project as a PhD candidate in Hill’s research group, says that efforts to develop treatments for preserving myelin have focused on cultivating young oligodendrocytes and protecting mature ones. But this study suggests the cells may change significantly as they age and that a one-size-fits-all treatment might not work.
“In response to the same thing, young cells go one way and old cells go another,” says Chapman, who is now a postdoctoral researcher at Stanford University. “If you wanted to protect the old cells, you may have to do something completely different than if you wanted to help the young cells mature. You’ll likely need a dual approach.”
The paper builds on a living-tissue model the team reported in the journal Nature Neuroscience in March 2023 that allows them to initiate the death of a single oligodendrocyte to observe how the cells around it react. They reported that when an oligodendrocyte in a young brain died, the cells around it immediately replenished the lost myelin. In a brain equivalent to that of a 60-year-old, however, the surrounding cells did nothing and the myelin was lost.
“That model gets us as close as we can get to the cell-death process that happens in the brain,” Hill says. “We’re able to model the effects of aging really well. Our ability to select a single oligodendrocyte, watch it die, and watch it regenerate or fail to regenerate allows us to understand what drives this process at the cellular level and how it can be controlled.”
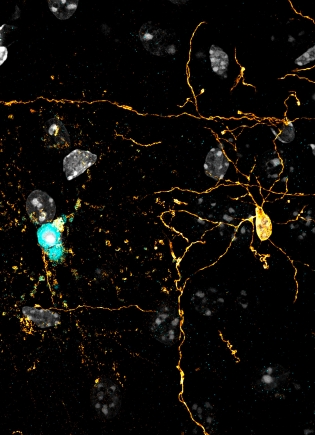
For the latest study, the researchers used their model to fatally damage oligodendrocyte DNA using what amounts to a cellular death ray—a photon-based device called 2Phatal that Hill developed. They also used the standard method for removing myelin that uses the copper-based toxin cuprizone as a comparison.
As previous studies have reported, the immature cells died quickly. But the older cells lived on, which the Dartmouth team at first interpreted as a resistance to DNA damage.
The study came into focus when the researchers examined the mature cells 45 days later using a long-term, high-resolution imaging technique developed in the Hill lab. “That’s when we saw that it wasn’t that the cells were resistant to damage—they were experiencing this extended cell death instead,” Hill says.
“No one’s ever checked for cell death that long after DNA damage. It’s the only example we can find in the literature where a cell experiences such a traumatic event and sticks around longer than a week,” he says.
Because humans have oligodendrocytes for life, the cells are known to accumulate DNA damage and be more resilient than other cells, Chapman says. “That’s why we think this effect is applicable to aging. One reason these cells may persist for such a long time is because they’re used to experiencing this kind of damage naturally in aging,” he says.
The study opens the first door of a vast labyrinth of more questions, Hill and Chapman say, such as whether the extended death is a good thing. It may be the equivalent of dysfunctional myelin, which is worse than if there was no myelin at all, Hill says. It isolates the cell from the surrounding tissue and essentially starves it of nutrients.
“It’s almost like there is garbage sitting on the axon for 45 days. Do we want to save that garbage or speed up its removal? We didn’t even know that was a question until we saw this,” Hill says.
“If we understand the cell-death mechanism, maybe we can speed it up and get rid of that dysfunctional myelin,” he says. “We’re always trying to save the cells and save the tissue, but you have to know if they’re worth saving.”